The short answer most people hear is “usually in the 30s or 40s.” That’s true for the first obvious symptoms, but it misses years—often decades—of quiet changes that start long before anyone trips, fumbles a sentence, or shows a visible movement. Today’s picture blends the classic timeline (adult onset with a juvenile form before 20) with newer biology that shows early synapse changes and a slow, DNA-level clock ticking inside vulnerable brain cells.
The basics: gene, risk, and the classic timeline
Huntington’s disease (HD) is caused by an expanded series of “CAG” repeats in the HTT gene and passes in an autosomal dominant pattern: each child of an affected parent faces a 50% chance of inheriting the expanded allele. Repeat length matters. Most people with ≥40 repeats develop HD during a normal lifespan; 36–39 repeats have reduced penetrance, and >60 often means juvenile onset.
Clinically, many first symptoms cluster in mid-adulthood: subtle executive problems (planning, multitasking), mood changes (irritability, depression), and small, dance-like movements called chorea. Some people first notice clumsiness or softer speech. The juvenile form looks different—more stiffness than chorea, school decline, and seizures.
What starts before symptoms: an early pruning problem
Work from Boston Children’s Hospital and Harvard Medical School has shown that two immune system players—complement proteins and microglia—switch on early in HD. They tag and prune synapses in corticostriatal circuits before motor signs appear. In mouse models, blocking the complement protein C1q prevented loss of these synapses and preserved thinking skills, suggesting an early therapeutic window. Levels of complement proteins were also higher in spinal fluid from people with HD even before motor diagnosis—hinting at a possible biomarker for “silent” disease activity.
This adds a practical message for families: even when movement looks normal, the brain may already be changing its wiring. That’s one reason research clinics track people who carry the gene years before they’re expected to show symptoms—looking for signals that could guide earlier treatment.
The slow DNA clock: somatic expansion and the 150-repeat “tipping point”
Another line of research explains why timing differs between people with the same inherited repeat length. Inside certain neurons, the CAG number doesn’t stay put. It expands over the years—a process called somatic expansion. A 2025 analysis of more than half a million single cells from human brains found that the neurons most at risk—striatal projection neurons—quietly add repeats across adulthood. Most cells look stable for decades, but once a neuron’s repeat length drifts toward ~150, its gene program falters; pass that mark and identity crumbles, “suicide” pathways turn on, and the cell dies within months. Meanwhile, neighboring neurons might still be far from that threshold. This asynchronous loss explains the gradual, stepwise decline families see.
If you inherit, say, 40 repeats, the model suggests a long first phase (years to add the first few dozen repeats), a faster middle stretch, and then a short final sprint once ~150 is reached. Not every cell follows the same calendar. That mosaic pattern—some neurons over the line, others not—also means treatments that slow expansion might still help even after symptoms begin.
Genetic “repair crew” proteins that monitor DNA mistakes can affect this pace. Variants in these pathways help explain why two siblings with the same baseline CAG length can have different ages of onset. Researchers now track somatic expansion in blood and spinal fluid alongside imaging—part of a wave of studies aimed at timing therapy better.
So… When does Huntington’s disease develop?
From birth—because the gene is there. From early adulthood—because microglia and complement begin to reshape synapses before anyone notices a tremor. And from the years leading up to diagnosis—because certain neurons are inching toward that ~150-repeat threshold that foretells cell death. Put simply: the clinical debut in the 30s or 40s is just the moment the invisible becomes visible.
What you can expect across stages
Premanifest (gene present, no diagnosis).
Thinking and mood may be normal, but research often finds subtle biomarker shifts—synaptic pruning signals, early imaging changes, and small rises in injury markers like neurofilament light in some studies. This stage can last decades.
Prodromal (subtle changes).
Family members may first notice short temper, less flexibility in routine, trouble with complex tasks, or small slips in balance. Many people still drive, work, and parent; targeted support and counseling start to matter a lot.
Manifest HD (diagnosable).
Movement disability grows, speech and swallowing need attention, and mood or motivation can sag. Most live 10–30 years after onset, with care needs rising over time. Pneumonia and falls are common hazards; planning ahead for nutrition, home safety, and support services pays off.
Testing, timing, and family planning
Predictive testing is available for adults with a parent who has HD. It’s a serious step, best taken with a genetic counselor and a clear plan for emotional, legal, and insurance implications. Some families consider IVF with preimplantation genetic testing to avoid passing on the expanded allele; others choose prenatal testing. There’s no single “right” path—only informed choices.
Treatment today: symptom control and function
There’s no approved drug that reverses the disease, but symptom care helps. VMAT2 inhibitors (tetrabenazine, deutetrabenazine) can reduce chorea; some antipsychotics and antidepressants support mood and behavior; and PT/OT/speech therapy, nutrition planning, and sleep strategies protect daily life. Care teams often include neurology, psychiatry/psychology, social work, and therapy services.
What’s new in trials: two big directions
1) Lowering toxic huntingtin.
Different approaches (antisense drugs, viral vectors delivering small RNAs) aim to reduce mutant huntingtin in the brain. In September 2025, multiple outlets reported early clinical data for AMT-130 (a one-time neurosurgical gene therapy) showing a ~75% slower clinical decline over three years in a small high-dose group compared with external controls, along with reduced neuron-injury markers. The data are preliminary and not yet fully published; larger studies and regulatory review are ahead, but this is a concrete step that patients and families have been waiting to see.
2) Slowing the DNA clock (somatic expansion).
Inspired by the human post-mortem cell studies, several teams are designing medicines to slow repeat growth inside neurons—potentially delaying the ~150-repeat tipping point. If this works, it could matter both before and after diagnosis, because not all neurons cross the line at once. This strategy complements huntingtin-lowering rather than replacing it.
What about the immune pruning story?
Because complement proteins appear to tag vulnerable synapses early, C1q-blocking antibodies are being tested. These aim to protect corticostriatal connections tied to movement and reward learning, where early loss appears to begin.
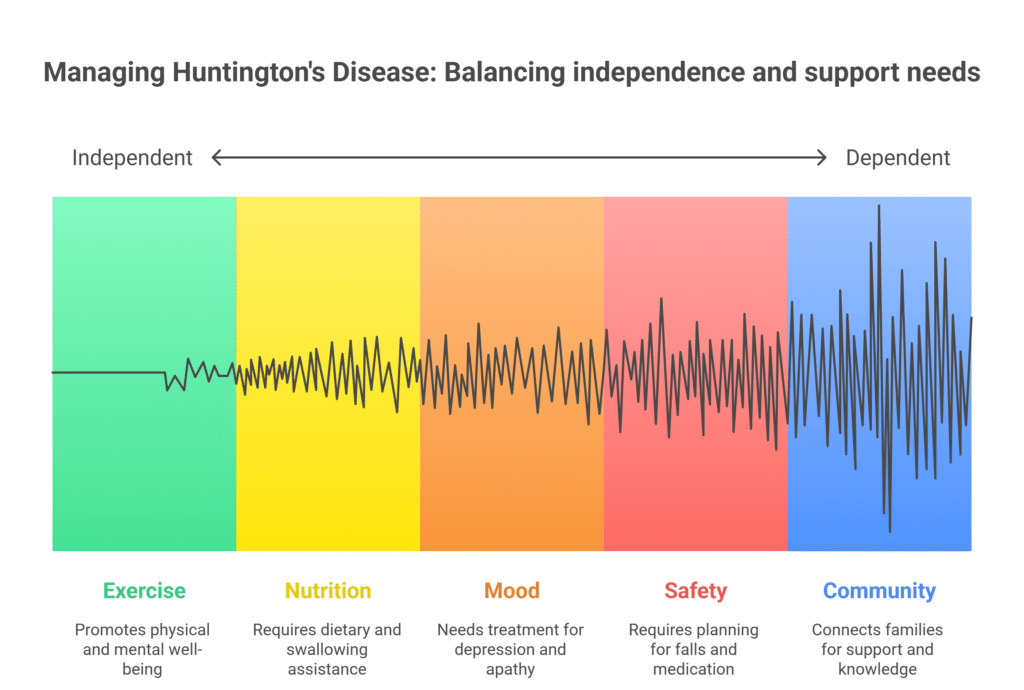
Living well in the meantime
- Exercise and activity. Regular, safe movement supports balance, mood, and sleep.
- Nutrition and swallowing. Calorie needs can soar when chorea burns energy; early input from dietitians and speech therapists helps.
- Mood and motivation. Depression and apathy are part of the illness, not a personal failing; treatment can help.
- Safety. Plan for falls, driving decisions, medication management, and home setup.
- Community. Local and online HD groups connect families with practical know-how and emotional support.
A timeline that finally fits what families see
Families often describe years of “something is off” before a formal diagnosis. The science now matches that lived reality. Early synapse pruning may trim the very circuits that control fine movement and flexible thinking, while a slow repeat-expansion clock pushes certain neurons toward a point of no return. Alongside classic genetics and inheritance, these two stories give clearer targets and better timing for care.
For now, if you carry the gene, it’s reasonable to think in layers of time:
- Today: symptom care, mental health, and support at home.
- This year: talk with your clinic about research registries and biomarker check-ins.
- This decade: watch the trial space—huntingtin-lowering, repeat-expansion modifiers, and complement-blocking drugs each attack a different part of the problem.
And never forget the simplest but most helpful actions: stay active, eat well, sleep as best you can, keep up with friends and hobbies, and line up the practical help you deserve. The science is finally describing what’s been happening all along, which puts earlier and smarter care within reach. When does Huntington’s disease develop? From gene to synapse to neuron, it’s a long road that begins quietly—but with growing tools to spot it early and, we hope, to slow it down.
Sources
- Overview and typical onset ages; juvenile HD: Mayo Clinic, Cleveland Clinic, NHS. Mayo Clinic+2Cleveland Clinic+2
- Inheritance and CAG ranges: GeneReviews. NCBI
- Early immune pruning and C1q blocking: Harvard Medical School / Boston Children’s. Harvard Medical School
- Somatic expansion and ~150-repeat threshold: Cell (2025) and explainer at HDBuzz. PubMed+1
- AMT-130 gene therapy reports (preliminary): ABC News and Sky News. ABC News+1
Note: News items on AMT-130 reflect early communications; peer-reviewed publications and regulatory assessments are pending.

